What is it?
December 27 marks the 192nd birthday of Louis Pasteur, which means that (a) he’d be really old if he hadn’t died in 1895, and (b) today is the perfect day to talk about tartaric acid.
Tartaric acid occurs naturally in many plants, particularly grapes. You’ve already read about ‘wine diamonds’ (potassium bitartrate), but you may not be aware of the contribution tartaric acid has made to scientific language.
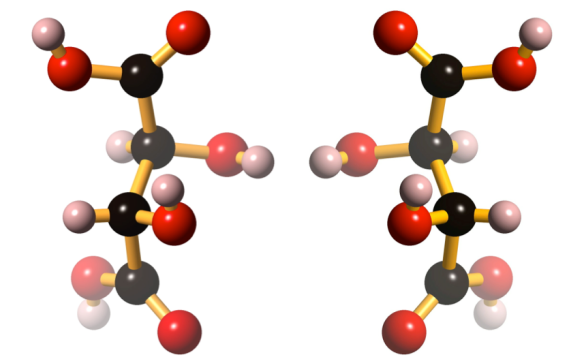
Naturally occurring tartaric acid, first isolated in 1769, was found to rotate plane polarized light to the right. When it was prepared synthetically, tartaric acid had identical properties, except that it didn’t rotate plane polarized light. The synthetic material was thought to be a different compound, and was named racemic acid (from racemus, Latin for ‘a bunch of grapes’). It was subsequently determined that tartaric acid can exist in two different forms; and that naturally occurring tartaric acid was L-tartaric acid, while ‘racemic acid’ was actually an equal mixture of D and L-tartaric acid, mirror image isomers (enantiomers). These enantiomers were optically active in opposing directions, appearing optically inactive; this explained the otherwise identical properties of tartaric acid and racemic acid.
For this reason, ‘racemic’ came to mean ‘an equal mixture of enantiomers’, and this term continues to be ubiquitous in organic chemistry today.
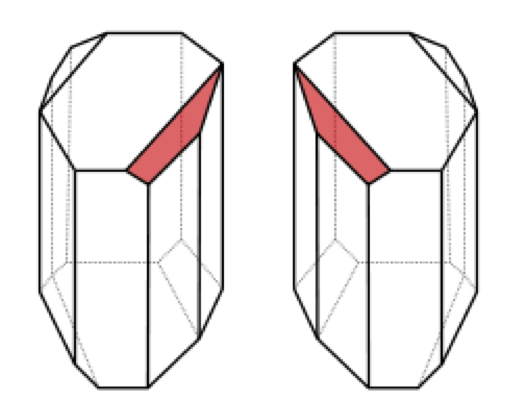
So where does Pasteur fit into this story? Early in his career, before he discovered vaccination, microbial fermentation and invented the process which still bears his name (pasteurization), Pasteur studied crystals of tartaric acid and ‘paratartaric acid’ obtained from wine sediments. In particular, he wondered why (as described above) tartaric acid rotated light, while paratartaric acid did not, even though the chemistry and elemental composition of the two were identical. In one of the most beautiful and famous experiments in the history of science, Pasteur noticed, while squinting down a microscope, that there were two subtlety different types of crystals in the samples of paratartaric acid, each the mirror image of the other (see diagram below). He very carefully (and tediously) separated the two types of crystals into separate piles, redissolved each pile, and found that each did indeed rotate light, but in opposite directions. He had, in effect, separated the two enantiomers from the paratartaric acid (a.k.a. racemic acid) and discovered molecular chirality.
What does it look like?
Where did the structure come from?
D-tartaric acid can be found under CCDC refcode TARTAC, while L-tartaric acid is at CCDC refcode TARTAL.