Jason Brouwer tells us about the killer protein he has been working on, at the Walter and Eliza Hall Institute, for his PhD.
It’s Halloween! And fittingly today’s crystal structure is the ‘Grim Reaper’ of cell biology, a protein called ‘Bak’.

Water-colour painting of the Grim Reaper by Rachel Bucknall, with the BH3:groove dimer structure of Bak as the face. The structure was generated using pymol.
Although you may not notice, billions of your cells die every day. This is a highly ordered process called apoptosis and is essential for the development and maintenance of our bodies. Apoptosis is the programmed death of stressed, damaged or unneeded cells. When it is disrupted it can result in the unwanted survival of cells, as in cancer, or unwanted loss of cells, as in degenerative diseases. Therefore we want to study exactly how cells die in order to further understand these diseases.
What does it look like?
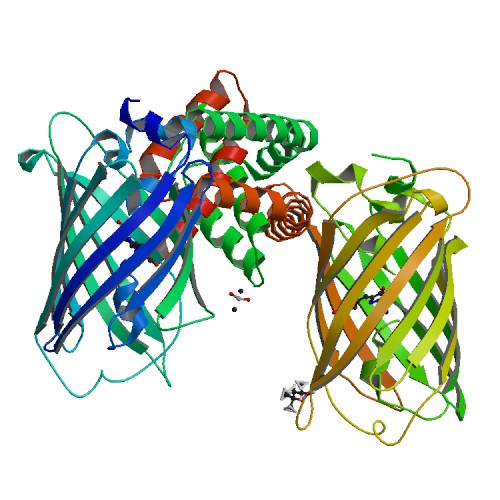
Biological assembly of Bak, generated by PISA. Image taken from the PDB http://www.rcsb.org/pdb/explore/explore.do?structureId=4U2V
What is it?
I study the proteins that control cell death, in particular the ‘killer protein’ Bak. Bak sits on the outside of the mitochondria, which are the energy factories of the cell. When the cell gets a signal to die, Bak is activated and interacts with other active Bak molecules to building large structures called oligomers that break open the mitochondria. This flood of contents from the mitochondria is a point of no return and the cell is committed to death. The crystal structure above captures a building block of the Bak oligomer, the BH3:groove dimer. The conserved BH3 domain of one Bak molecule sits in the hydrophobic groove of another. This forms a very strong, symmetric interaction that is essential for Bak mediated cell death.
The structure is from the publication Brouwer et al. in Mol Cell 2014 [55(6): 938-946], PDB accession code 4U2V. X-ray data was collected on the MX2 beam-line at the Australian Synchrotron.
